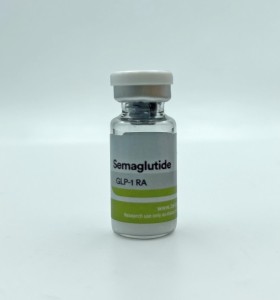
Semaglutide 5 Deus Medical
🔥 15% EXTRA DISCOUNT FOR DEUS MEDICAL 🔥
After adding the products to your cart, proceed to the 'shopping cart' section.
You can apply your discount by entering your coupon code in the 'Use Coupon Code' field.
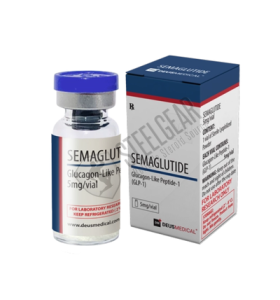

- Brand: Deus Medical
⚠️ WARNING: Potential Risks of Anabolic Steroids
Anabolic steroids can cause hormonal imbalances, liver strain, cardiovascular issues, and psychological effects, and should only be used under strict medical supervision. Unauthorized or improper use may lead to serious health complications.
If used, obtain these substances exclusively from licensed, verified medical providers to ensure safety and proper oversight.
SEMAGLUTIDE 5 mg Deus Medical
Important Disclaimer: The information provided in this article is intended strictly for general informational and scientific review purposes. SEMAGLUTIDE 5 mg by Deus Medical is discussed here exclusively from the perspective of an investigational "research compound." This text does not constitute definitive medical advice, nor does it guarantee specific health outcomes. Always conduct your own comprehensive research and consult with a licensed medical professional or endocrinologist before considering the use of any investigational supplement or peptide.
What is SEMAGLUTIDE 5 mg Deus Medical?
Semaglutide 5 mg by Deus Medical is a pharmaceutical-grade research peptide classified as a GLP-1 receptor agonist. Developed to replicate the action of the naturally occurring GLP-1 hormone, this compound is at the forefront of metabolic science.
While initially researched for Type 2 diabetes, Semaglutide has emerged as a groundbreaking agent in the study of chronic obesity management. By agonizing GLP-1 receptors located in the pancreas and the Central Nervous System (CNS), it provides researchers with a potent tool to observe improvements in glycemic index monitoring and weight reduction.
Technical Research Specifications
| Feature | Investigational Detail |
| Manufacturer | Deus Medical |
| Active Substance | Semaglutide |
| Concentration | 5 mg per vial |
| Pack Size | 1 Vial |
| Classification | GLP-1 Receptor Agonist |
| Form | Lyophilized Peptide (Injectable) |
Mechanism of Action: Physiological Effects
Research suggests that Semaglutide acts by triggering the body's natural metabolic responses when blood glucose levels rise. Current studies highlight four primary pathways:
Increased Insulin Secretion: It is hypothesized to stimulate the pancreas to release additional insulin in response to elevated blood sugar levels.
Lower Glucagon Levels: Observations indicate it decreases the secretion of glucagon, a hormone responsible for raising blood glucose, thereby assisting in glycemic control.
Delayed Gastric Emptying: By increasing the interval between food ingestion and evacuation into the small intestine, it produces a more stable glucose profile compared to other agonists.
Neuro-Satiety Signaling: It interacts with the brain’s feeding centers, theorized to help subjects feel fuller with smaller portions, thereby reducing excessive caloric intake.
Investigational Benefits
Semaglutide 5 mg is extensively studied for the following potential therapeutic benefits:
Glycemic Optimization: Clinical research demonstrates a remarkable improvement in HbA1c levels, supporting long-term blood sugar stability.
Profound Weight Loss: As a major innovation in obesity research, the compound is studied for its ability to help subjects shed significant amounts of weight.
Convenient Dosing Schedule: Unlike daily metabolic agents, Semaglutide is researched as a once-weekly protocol, which significantly improves adherence in clinical studies.
Possible Side Effects & Safety Profile
Even within controlled research settings, potential side effects must be monitored:
Gastrointestinal Sensitivity: Diarrhea, constipation, and vomiting are the most common initial reactions, usually occurring during the induction phase.
Hypoglycemia Risks: While rare, low blood sugar can occur, especially if the subject is utilizing other diabetes-related medications like insulin.
Thyroid Monitoring: Animal studies have suggested a potential risk of thyroid C-cell tumors; therefore, subjects are typically kept under observation for any thyroid gland abnormalities.
Frequently Asked Questions (FAQ)
1. Who is the primary subject for Semaglutide research?
Research primarily focuses on individuals with Type 2 diabetes or those classified as obese/overweight. Investigational use must always be guided by a subject's medical history and professional clinical oversight.
2. How is Semaglutide 5 mg typically administered in studies?
The compound is administered subcutaneously once per week. Common injection sites for research include the abdominal wall, thigh, or upper arm.
3. Is Semaglutide compatible with other research agents?
Yes, it is often studied alongside other antidiabetic medications (like linagliptin), though extreme care regarding possible interactions is required.





















 Ask us anything on Telegram!
Ask us anything on Telegram!